|
11/23/2023 0 Comments Na3po4 and nacl precipitate color 
Pat yourself on the back, you’ve just balanced a net ionic rxn. Balance your net ionic reaction and add states of matter. 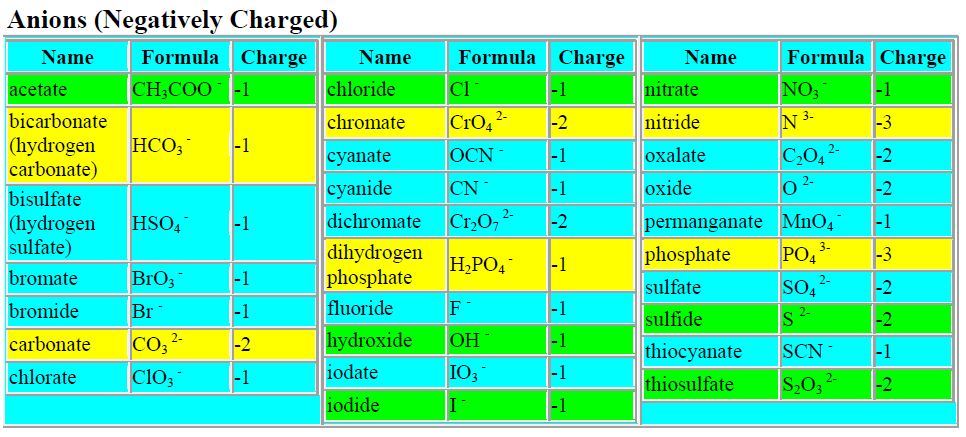
Write down the insoluble product, then list the ions that had to form it as reactants. In net ionic reactions, only non-spectator species are actually shown because they’re the only ones doing anything observable. A student wants to determine if PO43 is part of. Sodium phosphate reacts with calcium chloride resulting in the formation of calcium phosphate and sodium chloride. BaCl2 (aq) + Na2SO4 (aq) 2 NaCl (aq) + BaSO4 (s) The ions that remain in solution (don’t form a precipitate) are called “spectator ions.” In rxn 1, the spectators are Na+1 and NO3-1 In rxn 2, all of the ions are spectators…hence ‘no reaction’ In rxn 3, the spectators are Na+1 and Cl-1Ĥ Net Ionic Reactions Something to think about…When you get a paycheck, you’re more interested in the net pay than the overall pay because the net pay is the most inherently interesting. Mg3(PO4)2 MgPO4 Na Mg O NaCl NaCl2 QUESTION 8 Solutions of ZnCl2 and Na3PO4 are mixed and a precipitate forms. Sodium phosphate reacts with hydrochloric acid to form phosphoric acid and sodium chloride. How about: 2 NaCl (aq) + Pb(NO3)2 (aq) NaNO3 (aq) + PbCl2 (s) 2 Yes, because a precipitate was formed.Ģ Solubility Rules Soluble (will NOT form ppt’s)Īll nitrates, acetates, ammonium and Group 1 salts All chlorides, bromides and iodides except Ag, Pb and Hg1+ All fluorides except Group 2, Pb2+, Fe2+ All sulfates except Ca, Sr, Ba, Hg, Pb2+, and Ag Insoluble (WILL form ppts) All carbonates and phosphates except Group 1 and ammonium All hydroxides except Group 1, Sr and Ba All sulfides except Group 1, 2 and ammonium All oxides except Group 1ģ Using your solubility rules chart predict the products for the following double-replacement reactions. Start with the most complicated formula, 'Ca'3('PO'4)2. Consider: 2 NaCl (aq) + Mg(NO3)2 (aq) NaNO3 (aq) + MgCl2 (aq) 2 Did a reaction actually occur? (Hint: Think about the 5 indicators of chemical change) Energy released Gas evolution Color change Temperature change Precipitate formation No, there is no reaction because all of the ions remain in solution. Your unbalanced equation is 'Na'3'PO'4 + 'CaCl'2 'Ca'3('PO'4)2 + 'NaCl' 1. The solubility rules allow us to determine if a double-replacement reaction will actually occur. Place 2 ml of 0.1 M Na3PO4 solution in a test tube and 2 ml of your unknown. You can use ammonia solution instead of sodium hydroxide solution, but there are different results for aluminium and copper(II) salts when you use excess ammonia.1 NaCl (aq) + Mg(NO3)2 (aq) NaNO3 (aq) + MgCl2 (aq) 2 The color for unknown B was clean while the color of NaCl was a milky color. Solutions containing copper(II) ions form a blue precipitate when mixed with sodium hydroxide solution. Solutions containing copper(II) ions form a blue precipitate when mixed with sodium hydroxide solution add dilute sodium hydroxide solution until it is in excess and record the result.Fizz (effervescence), aroma, color change, temperature change, solid (precipitate) and new substance. record the colour of any precipitate that’s formed. Solve: CaCl2 + Na3PO4 -> Ca3(PO4)2 + NaCl. Will a precipitate form when 1.00 g of AgNO3 is added to 50.0 mL of 0.050 M NaCl If so, would you expect the precipitate to be visible The likely precipitate formed by mixing BaCl2 and AgNO3 would have the formula: a) No precipitate will be formed.add a few drops of sodium hydroxide solution.place about 5cm 3 of the solution into a test tube.dissolve a small quantity of the substance in water.When metal ions combine with the hydroxide ions (OH - ) from either sodium hydroxide solution or ammonia solution, they form insoluble precipitates with characteristic colours. Many tests for anions and cations are precipitation reactions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
